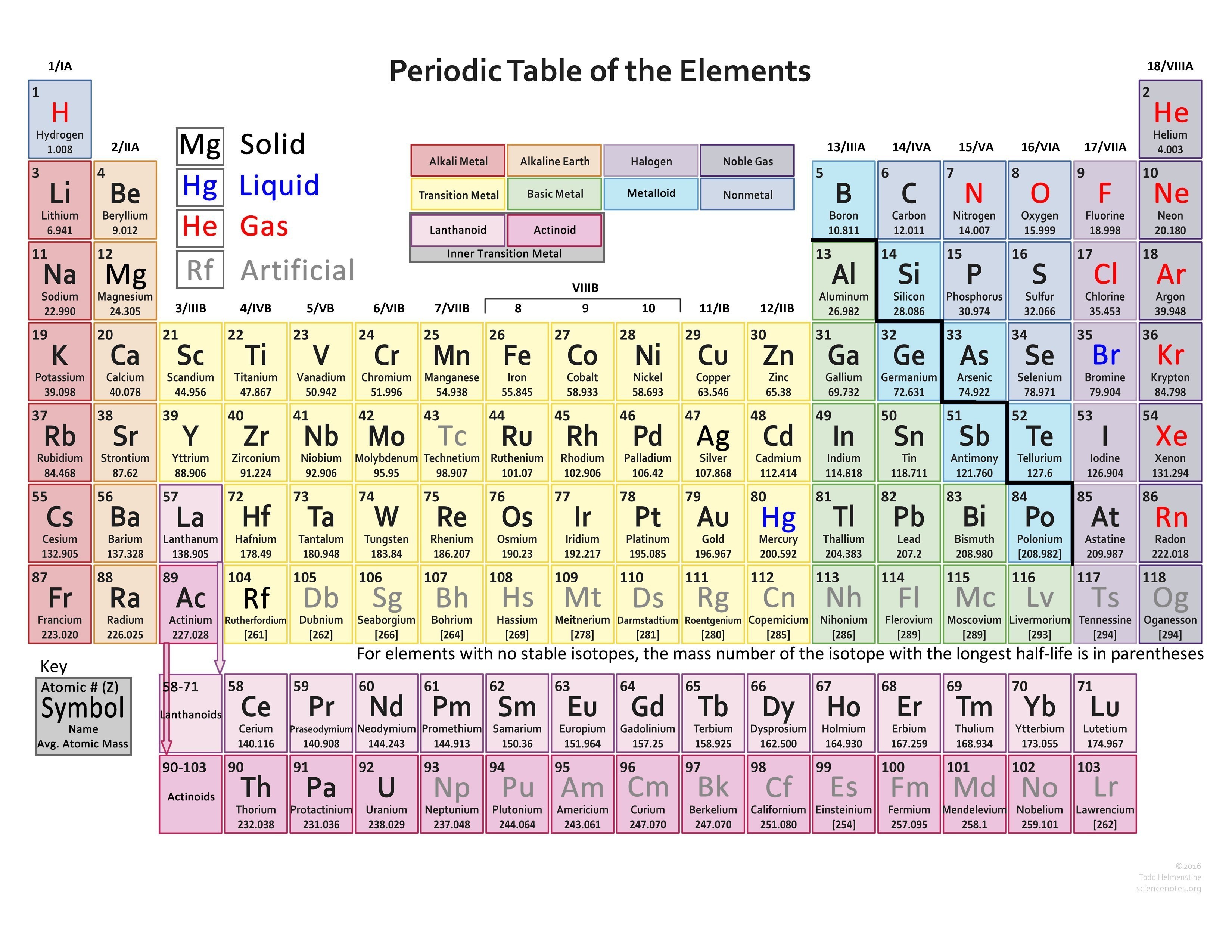 
For example the far right-hand column contains gases which are all chemically inert. Originally the elements were arranged on the periodic table, from left to right, according to their atomic number, and in columns according to their chemical and physical properties. The periodic table reflects this situation. This is a statement of what is called the octet rule. so that each atom ends up with 8 electrons in their valence shell. They do this by reacting with one another to gain, lose or share electrons. Chemistry can be thought of as basically a consequence of all of the elements on the periodic table trying to achieve the same number of valence electrons as one of the inert gases. All of the other elements on the periodic table would like to be like an inert gas in terms of the number of electrons they have in their valence shell. The inert gases are called inert because they are very unreactive with themselves and other elements. Even if an energy level can hold more than 8 electrons, the most stable number, is 8. (Each energy level can contain up to 2 n electrons, where n is the energy level refer to Section 3.2 and Table 3.4 in Raymond). This is because the first energy level can only hold 2 electrons, whereas the other energy levels can hold 8 or more electrons. All of the inert gases have 8 valence electrons, except helium, which has 2. 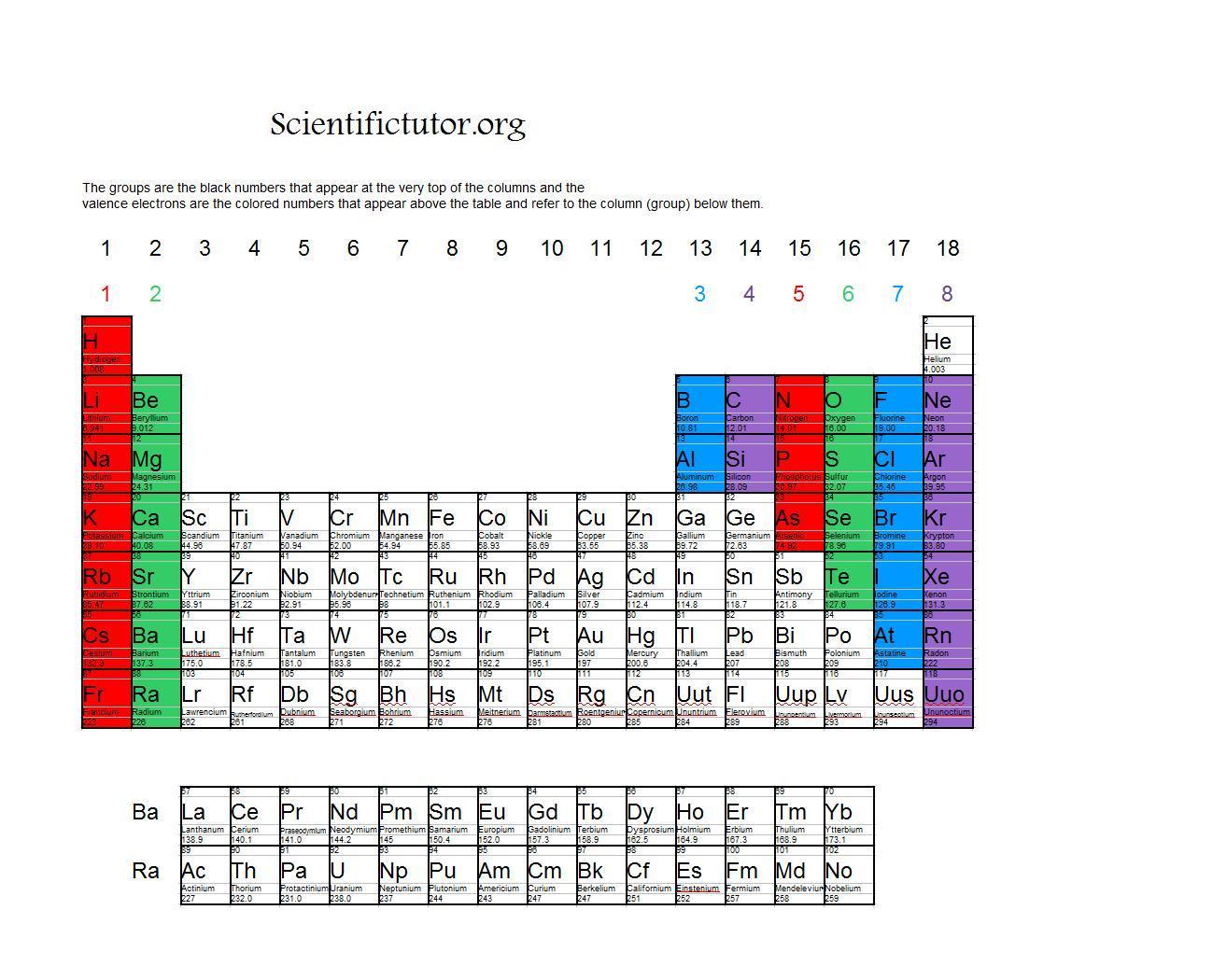
These elements, as a group, are are called the inert gases. The elements that have this number in their pure neutral forms are the elements in Group VIIIA. This happens be the most stable situation for an atom. 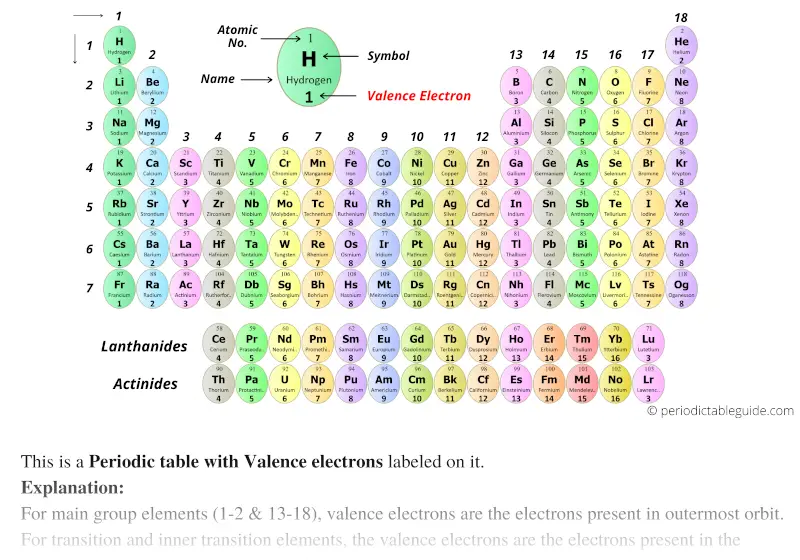
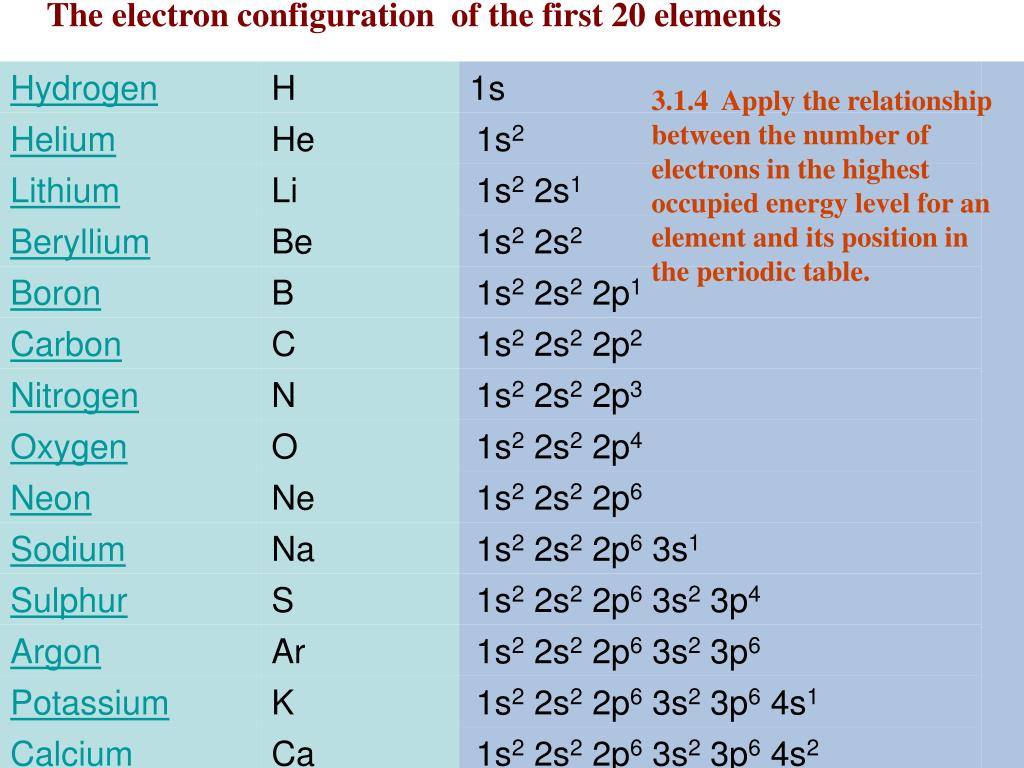
It turns out that there is something special in nature about having eight electrons in the valence shell. In the electron dot structures used here, valence electrons are shown as dots. Representative elements in the same group have the same number of valence electrons. Note that the number of valence electrons that each element has is equal to its group number, e.g., elements in Group IA have one valence electron (one dot), elements in Group IIA have two valence electrons (2 dots), etc.įigure 1 ( Figure 3.6 from Raymond ): Valence Electrons. Figure 3.6 in Raymond uses Lewis electron dot structures to show the number of valence electrons in some of the representative elements. Together, these elements are referred to as the main group or representative elements. For our discussions we will focus on the elements in the first two columns on the left-hand side of the periodic table and the last six columns on the right-hand side of the table. These are the electrons found in the highest occupied energy level or shell for an atom of an element. The Arabic numbering system is the most widely accepted today.In Section 3.2 the concept of valence electrons was introduced. Many periodic tables include both Roman and Arabic numbers. The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right.The CAS system used letters to differentiate main group (A) and transition (B) elements.The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.Three systems have been used to number families and groups: Recognizing Families on the Periodic TableĬolumns of the periodic table typically mark groups or families. Noble Gases: - Group 18 (VIIIA) - 8 valence electrons.Halogens: - Group 17 (VIIA) - 7 valence electrons.Oxygen Group or Chalcogens: - Group 16 (VIA) - 6 valence electrons.Nitrogen Group or Pnictogens: - Group 15 (VA) - 5 valence electrons.Carbon Group or Tetrels: - Group 14 (IVA) - 4 valence electrons.Boron Group or Earth Metals: Group 13 (IIIA) - 3 valence electrons.Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons.Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons.Alkali Metals: Group 1 (IA) - 1 valence electron.Many chemists and chemistry textbooks recognize five main families:Īnother common method of categorization recognizes nine element families: However, there are different ways of categorizing elements into families. Because element properties are largely determined by the behavior of valence electrons, families and groups may be the same. Element groups, on the other hand, are collections of elements categorized according to similar properties. The characteristics of the elements in these families are determined primarily by the number of electrons in the outer energy shell. Elements are classified into families because the three main categories of elements (metals, nonmetals, and semimetals) are very broad. Element families are indicated by numbers located at the top of the periodic table.Īn element family is a set of elements sharing common properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
